Potential energy is the energy of position
The concept of potential energy (PE) can be a little difficult to wrap your mind around.
Kinetic energy (KE) is easier: Things that are moving obviously have energy. A baseball can knock you on the head (and hurt), a moving train could flatten you, and so on. But an object (stationary or moving) also has a kind of energy just by virtue of where it is located with respect to other objects in the universe, and that may seem a little weird at first.
The first, most obvious example is that of gravitational potential energy energy.
A baseball on flat ground has nowhere to go, but a baseball lifted in the air or located on a hill has the potential to fall or roll – to move under the influence of gravity. That movement is an expression of KE that used to be stored as PE.
Forces are the reality that create the concept of potential energy. Without forces, there would be no potential energy. The examples below illustrate the concept of the force field, a sort of map of how invisible forces between objects change as their relative position changes. The force field, forces and the potential energy are related.
Here are a few examples of how potential energy is created by doing work.
Examples
- An object is raised against the force of gravity.
- A spring is stretched or compressed from its natural length.
- The string of a bow is pulled, flexing the limbs of the bow.
- The elastic bands of a slingshot are stretched backward before a shot.
- Air is pressurized so that it can expand against a piston and operate the air brakes on a truck.
- The level of water is raised behind a dam so that it can fall a long distance and generate electricity.
- A potential energy difference is created in a battery by charging it.
Gravitational potential energy
Gravity is a force that we can model mathematically very well using the universal law of gravitation,
$$F_g = \frac{G m_1 m_2}{r^2}$$
where G is the gravitational constant,
$$G = 6.674 \times 10^{-11} \frac{m^3}{s^2 K},$$
$m_1$ and $m_2$ are the masses of two objects, and $r$ is the distance between them in meters. Any object with mass exerts a gravitational force on any other object with mass. Planets and stars, of course, are very large (massive), thus they exert the largest forces.
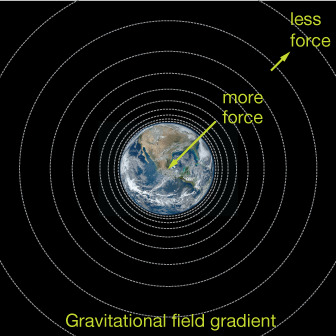
The gravitational force produced by any thing on another thing is inversely proportional to the square of the distance between (see box below). That means that if I double my distance from the center of Earth, the gravitational attraction that Earth and I exert on each other is reduced to ¼ its original value.
In the diagram, the circles represent spheres of constant force. The closer the spacing between circles, the greater the force. In this way, the potential energy has the look of a topographic map: the closer the curves, the steeper the slope of the hill.
According to universal law of gravity, the gravitational force between to objects is
$$F_g = \frac{G m_1 m_2}{r^2}$$
where $G$ is the gravitational constant, $m_1$ and $m_2$ are the masses of the two objects and $r$ is the distance between them. If our initial distance is 1, then $F = G m_1 m_2.$ If we double that distance to 2, then because 22 = 4, the force becomes $F = \frac{1}{4} G m_1 m_2$
Gravitational potential "well"
This image is a representation of the gravitational potential energy around a planet. The planet would lie at the center of the "well".
At far distances where the gravitational force is weak, the potential well is not steep, and the force is small. Near the planet the surface becomes much steeper. The force felt by an object attracted to the planet is proportional to the steepness of this potential. In fact, force is precisely the slope of this 3-D curve in a given direction.
If the graph represents the potential energy function, then its steepness (its derivative in calculus) at any point is the gravitational force at that point.
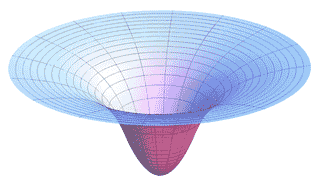
Albert Einstein showed that space (or "space-time") can actually be viewed as a 2-dimensional surface like this, and that the masses of the planets and stars warp that surface as shown in the figure. Objects then "fall" into that potential energy "well."
The pendulum
The pendulum is a great example of how gravitational potential energy works. Here we can toss aside the warping of space-time and just think of gravity as an invisible downward force.
The pendulum works solely because of the force of gravity.

When the weight of the pendulum is raised, we do work to give it potential energy that is proportional to the height. At the endpoints of the arc of the pendulum weight, there is a moment as it changes direction when its velocity is zero, so it has no kinetic energy at all. At those points, all of its energy is potential energy.
At the bottom of the swing, when the weight points toward the center of Earth, it has no potential energy relative to where it started. It is, however, moving as fast as it will ever move, all of its PE having been converted to KE.
Then the process begins all over again, as the weight rises, it loses speed (and thus KE), and gains gravitational PE. The only thing that slows the whole thing down is a little bit of friction with air molecules that eventually will cause the pendulum to stop.
Potential energy is a storage mode for kinetic energy. As a pendulum moves from side to side, all of its KE is converted to PE momentarily, only to be converted back to KE, and so on.
Elastic potential energy
Many devices can store elastic potential energy and release it as kinetic energy: a spring, a rubber band, an archer's bow, a catapult ...
The animation on the right shows a compressed spring at rest. In its compressed state, the spring stores elastic potential energy. The downward force it exerts on the hanging mass is proportional to the amount of compression (see Hooke's law).
Once the mass reaches the middle of its travel, and is at its equilibrium (unstretched / uncom-pressed) length, it will then begin to be stretched, storing more potential energy in that way. The force exerted on the hanging mass is greatest at either end, less in the middle, thus the mass has its greatest elastic potential energy when the spring is most compressed or most stretched.
Of course, it's also possible to over-stretch a spring, ruining its energy storage properties. My students do that with Slinkies™. I don't know why.
The potential energy of a spring is given by
$$PE = \frac{1}{2} kx^2$$
where k is a constant that depends on the propoerties of the particular sping and x is its stretched or compressed length from its equilibrium length.
Electrostatic potential energy
Charged objects, like electrons (-) and protons (+) exert invisible forces on one-another. These are called electrostatic forces. Like gravity, these are non-contact forces (one object doesn't have to touch another in order to exert a force on it). But unlike gravity, electrostatic forces can be attractive or repulsive. (Gravity is always attractive — no one ever just gets ejected off the planet while walking down the street.)
Here is a sketch of oppositely-charged particles in close approach. The circles represent "lines of force." Just like in our gravity example above, the closer together the lines of force, the more force exerted on and by the particles. This is always the case with oppositely-charged particles: The electrostatic force causes oppositely-charged particles to attract.
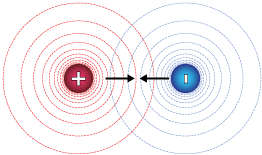
If we looked at these lines of force in three dimensions (think of them again as a topographic map), the red circles might extend above the page, and the blue ones below — the difference between positive and negative forces. The lines of force for like-charged particles, such as two electrons or two protons, looks like this.

These particles repel one another. In 3-D, these lines of force would both form mountains below the plane of the page.
Coulomb's law
The force between two charged particles is given by Coulomb's law:
$$F_{es} = \frac{k q_1 q_2}{r^2}$$
where k is a constant called the Coulomb constant (k = 8.98 × 109 N·m2·C-2), q1 and q2 are the two charges (in Coulombs, C), and r is the distance between them in meters. Notice how similar Coulomb's law is to the universal law of graviation. Both are inverse-square laws because the force is inversely proportional to the square of the distance between the particles.
Remember that with an inverse-square law, the force drops off with the square of the increased distance. For example, if the distance is doubled, the force falls to ¼ of its previous value.
Calculating gravitational PE & the units of potential energy
Gravitational potential energy (PE) is easy to calculate, and we can use it to get an idea of the usefulness of PE.
It turns out that the potential energy gained by raising a mass, m, to a height h is exactly equal to the amount of work needed to lift it there, which is the force (mg, where g = 9.8 m·s-2, is the acceleration of gravity) multiplied by the distance moved (h).
$$PE = m g h$$
Units of PE, KE, work
The unit of potential energy (PE), kinetic energy (KE) or work is the Joule.
$$ 1 \: \text{Joule (J)} = 1 \: \frac{Kg \cdot m^2}{s^2}$$
Example 1
How much potential energy does a 46 g golf ball have if elevated to the level of the observation deck (381 m) of the Empire State Building? Given that the kinetic energy of the ball will be $KE = \frac{1}{2} mv^2$ just before it hits the street after being dropped, how fast will the ball be falling, and is this reasonable?
$$ \begin{align} PE &= mgh \\[5pt] &= (0.046 \, Kg)\left( 9.81 \, \frac{m}{s^2} \right) (381 \, m) \\[5pt] &= 172 \, J \end{align}$$
Now energy is conserved, so we expect that all of this potential energy will be converted to kinetic energy by the time the ball is at street level. To calculate that velocity, first rearrange $KE = \frac{1}{2} mv^2$
$$KE = \frac{1}{2} mv^2$$
$$ \begin{align} 2 \, KE &= mv^2 \: \: \color{#E90F89}{\leftarrow \: \text{multiply by 2}} \\[5pt] \frac{2KE}{m} &= v^2 \: \: \color{#E90F89}{\leftarrow \: \text{divide by m}} \\[5pt] v &= \sqrt{\frac{2 \, KE}{m}} \: \: \color{#E90F89}{\leftarrow \: \text{square root}} \\[5pt] \end{align}$$
Now we have
$$v = \sqrt{\frac{2 (172 \, J)}{0.046 \, Kg}} = 86 \, \text{m/s}$$
Well, that's a very fast speed – about 192 mi./h. That's not too likely. The ball would encounter increasing air resistance on its way down as it banged into air molecules. These collisions would slow it down, eventually resulting in a steady velocity ("terminal velocity") of about 32 m/s.
Example 2
A certain recurve bow takes 80 lbs. of force to pull an arrow back by about 70 cm. How much potential energy will be stored in the bow before the arrow is shot? (1 lb. = 4.448 Newtons).
$$ \begin{align} \require{cancel} w = PE &= F·d \\[5pt] &= 80 \cancel{lbs.} \left( \frac{4.448 \, N}{1 \, \cancel{lb}} \right)(0.70 \, m) \\[5pt] &= 249 \, J \end{align}$$
Work is done on the bow to store potential energy in its flexible arms. When the arrow is released, that PE is converted into kinetic energy of the bow arms and string, which, in turn, propels the arrow forward. Most of the PE ends up in the arrow, propelling it with high velocity. Like in example 1, you could calculate an upper limit on the speed of the arrow. Give it a try; assume that a typical target-practice arrow weighs 19 g.
Example 3
How much potential energy does a 50 Kg cart have (relative to being on level ground) after having been pushed up a 5˚ ramp to a height of 5 m ? Would there be any difference in PE gained if the cart was simply lifted to a height of 5 m?
$$ \begin{align} PE &= mgh \\[5pt] &= (50 \, Kg)\left( 9.81 \frac{m}{s^2} \right)(5 \, m) \\[5pt] &= 2,452 \, J \: \text{ or } \: 2.45 \, KJ. \end{align}$$
The potential energy would be the same if the cart was just lifted straight up to a height of 5 m. Work and PE are known as "state functions." They only depend on the final and initial conditions (i.e. final and initial KE), and not on how the system got from one to the other.
Force & potential energy and caclulus
Force and potential energy (the potential energy function) are related through the derivative in calculus. Force is the slope of a one-dimensional potential function (like a mass on a spring). Force can also be a slope in any direction or a gradient on a multi-dimensional potential energy surface. In the one-dimensional case,
$$F(x) = \frac{d}{dx} V(x),$$
where $V(x)$ is the potential energy function (often just the "potential function" or "the potential."
Likewise, the total work done or potential energy gained by moving an object a distance x under a force F is the integral of the force over the distance:
$$w = PE = \int_a^b \, F(x) dx$$
Example: Spring potential
The simplest potential function representing the stretching and compressing of a spring is the harmonic potential,
$$V(x) = \frac{1}{2} kx^2,$$
where k, the spring constant, is a characteristic of the spring, it's composition and how it is formed. The potential is just a parabola like this one, in which k has been set to 1.
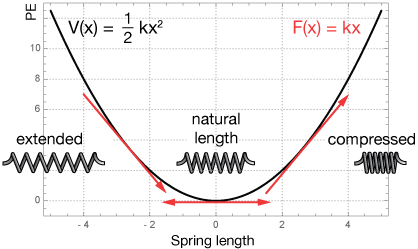
Under this potential, the restoring force of the spring is negative when the spring is stretched and positive when it is compressed, by convention. The force increases quadratically with the extent of the stretching or compressing. We calculate the force at any point by taking the derivative of the potential at that point:
$$F(x) = \frac{d}{dx} V(x) = kx$$
The red arrows in the potential plot are tangents at three select points showing negative and positive restoring forces, and the horizontal tangent at the natural length of the spring, where no restoring force is required.
SI units
SI stands for Système international (of units). In 1960, the SI system of units was published as a guide to the preferred units to use for a variety of quantities. Here are some common SI units
| length | meter | (m) |
| mass | Kilogram | (Kg) |
| time | second | (s) |
| force | Newton | N |
| energy | Joule | J |
xaktly.com by Dr. Jeff Cruzan is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. © 2012-2025, Jeff Cruzan. All text and images on this website not specifically attributed to another source were created by me and I reserve all rights as to their use. Any opinions expressed on this website are entirely mine, and do not necessarily reflect the views of any of my employers. Please feel free to send any questions or comments to jeff.cruzan@verizon.net.
