From atomic orbitals to molecular orbitals
By now you've spent some time learning about how electrons are arranged in atoms into atomic orbitals. Now it shouldn't be surprising that when atoms come close enough to share electrons in covalent bonds, those orbitals change their shapes. After all, it is the process of being bound to a nucleus that creates the atomic orbitals that are the bound electrons. The close proximity of another nucleus from a neighboring atom must also have a profound effect.
The mathematics of quantum mechanics tell us that the atomic orbitals of individual atoms "mix" (in very specific, mathematically defined ways) to form new orbitals we'll call molecular orbitals.
In this section we'll mainly look at the carbon atom and the compounds it can form with other carbon and hydrogen atoms (hydrocarbons). This will give us a pretty complete view of these rearrangements of atomic orbitals into "hybrid" molecular orbitals.
We'll look here only at what we call "open-shell" interactions, bonds that form between two or more atoms with incomplete valence shells, such as the four single bonds formed between a carbon atom and four hydrogen atoms to form methane (CH4).
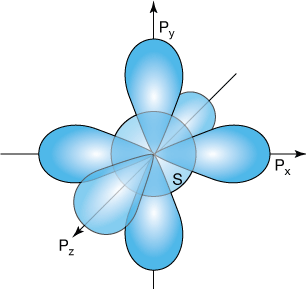
Pro tip:
Always remember, that even though we often speak of electrons "filling" orbitals, the orbitals actually are the electrons. A 3-D orbital does not exist until it is formed by an electron bound to a nucleus, or in the case of molecules, a group of nuclei.
Formation of molecular orbitals
We won't be able to dive into the complete mathematics here – you'll need to learn a little more math for that (don't worry, you can do it) – but we can illustrate the results.
Let's first think about two hydrogen atoms, each consisting of a proton and one electron – the simplest neutral atoms with an open valence shell. We'll think about how these atoms form a bond. First we ought to think about why they form a bond. The answer has three parts:
- The valence shell of each atom is incomplete, so according to the Pauli exclusion principle, there is room for one more electron in each, and we know that a full shell is more energetically stable,
- At short distances, the electron of each atom is attracted to the nucleus of the other atom, and
- At very short distances the electron clouds begin to overlap and repel.
Any chemical bond arises from a combination of sufficient attraction and strong overlap repulsion.
Now when we bring two H atoms together to form H2, we're bringing two atomic orbitals together. The laws of quantum mechanics tell us that when we "mix" two atomic orbitals (two s-orbitals in this case) we have to produce two molecular orbitals.
In terms of energy, these look like this:
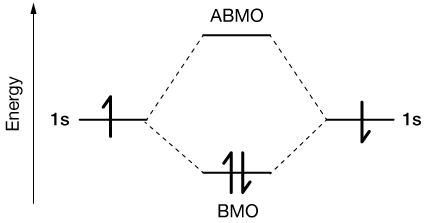
Each of the 1s orbitals of the H atoms has mixed into two molecular orbitals. One is of lower energy than the individual 1s energy levels and is called the bonding molecular orbital (BMO). The other is of higher energy. If either of the electrons in a bonding MO are excited into it (the anti-bonding MO, ABMO), the molecule flies apart into two atoms once again. The energy spacings here are just schematic, but the idea is the same for any bond. You can look at it like a conservation of orbitals. N atomic orbitals will always combine into N molecular orbitals, some bonding and some anti-bonding.
Pauli exclusion principle
The Pauli principle states that no two electrons bound in an atom or molecule may have exactly the same set of quantum numbers, n, L, mL and ms. It is the reason that each orbital can hold no more than two electrons, and that each of those has opposite spin.
Formation of a bond from two atomic orbitals means "mixing" those orbitals according to the rules of quantum mechanics, which results in two molecular orbitals (MOs), a bonding MO (BMO) and an anti-bonding MO (ABMO).
When carbon bonds, it forms hybrid orbitals
A neutral carbon atom in its ground state has four electrons in its outer (n = 2) shell, two electrons in the 2s orbital and two in the 2p orbitals. Four valence electrons means (among other scenarios we'll get to) four possible single bonds to a single carbon. For the moment, we'll think about methane, CH4, for simplicity.
Now those four electrons are in different kinds of orbitals (s and p), but we know from experiments that the four C–H bonds of CH4 are equivalent. It turns out that nature creates hybrid orbitals, consisting of three 2p orbitals and one 2s orbital, called sp3 orbitals (one part s, three parts p)
The diagram below shows how it works. After hybridization, all four valence electrons of the carbon atom occupy equivalent sp3 hybrid orbitals, ready to bond to four hydrogen atoms.
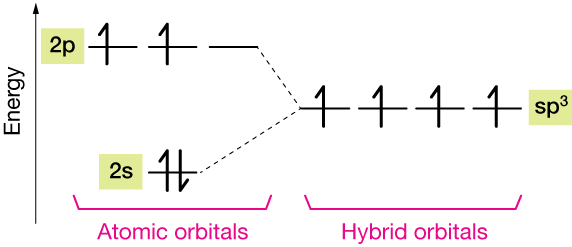
The diagram below shows the approximate difference between the atomic and hybrid orbitals of a carbon atom.

Valence shell electron-pair repulsion (VSEPR) theory predicts that the four main lobes of each of the four hybrid orbitals will move as far from one another as possible. That means they'll assume the tetrahedral arrangement shown above. Ultimately, this leads to the tetrahedral structure of CH4, and to the fact that most carbon "centers" in organic (carbon containing) molecules are tetrahedral.
The bonding and structure of methane, CH4
Now the molecular orbitals of methane form from the four 1s orbitals of each hydrogen atom and each of the four hybrid orbitals. Here's a schematic diagram of the approach of the hydrogens.
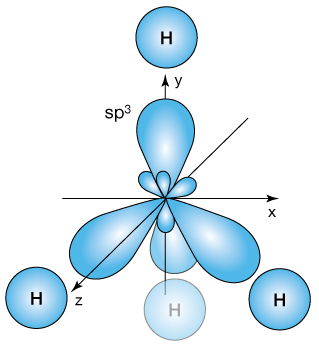
As each hydrogen bonds, an sp3 orbital of the carbon mixes with a hydrogen 1s orbital to create a bonding molecular orbital (BMO) and an anti-bonding orbital (ABMO). So long as the two electrons forming the bond occupy the BMO – the lowest energy state, a bond exists.
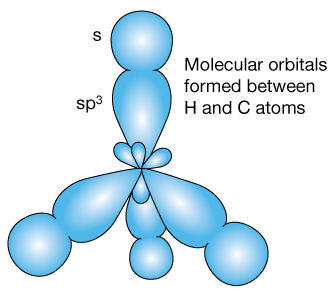
The fully bonded structure might look something like this. VSEPR theory explains the tetrahedral arrangement of the H atoms around the central carbon: The electron-dense orbitals move under the force of like-charge repulsion to create the largest-possible separation.
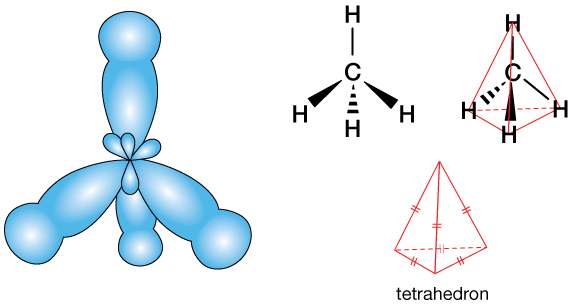
Except where double or triple bonds exist in molecules, every carbon atom is a tetrahedral center, with just such a structure, because of sp3 orbital hybridization. The figure below depicts one of the structures of glucose. The five carbon atoms in
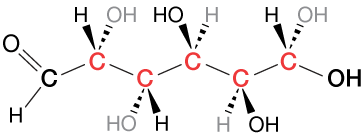
Chirality
One of the most interesting implications of this embedded tetrahedral structure in carbon-containing molecules is that of chirality, or handedness. We can, in fact, have right-handed and left-handed molecules. Take a look at the two chiral centers below. They are mirror images of each other. From the point of view of the ligand labeled "1" in each, counting around ligands 2, 3 and 4 produces a clockwise circle on the left and a counterclockwise circle on the right. This actually has a huge effect on the chemistry of carbon, including all of biochemistry – but it's a subject for another section.
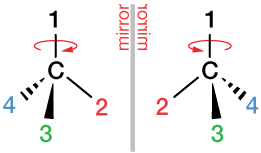
ligand
A ligand is anything that bonds to an atom or molecule of interest. Which atom or molecule is in the context of the statement. The four hydrogen atoms of CH4 are ligands of the carbon atom. An oxygen molecule, O2, is a ligand for the heme group in hemoglobin, and so on.
C=C double bonds and sp2 hybridization
Now we turn to a second hybrid orbital, this one composed of one part s-orbital and two parts p orbital. The figure below shows the energy-level diagrams that lead to such hybrid orbitals.
Notice that in this case, hybridization first requires excitation of one electron (up and down arrows in the figure) from the s-orbital to a p-orbital first.
These sp2 hybrid orbitals will give rise to a new kind of bonding. You know it as double bonding, but these bonds are also called π-bonds, while single bonds are called σ-bonds.

The resulting sp2 orbitals arrange themselves around the 2p orbital as shown below, with 120˚ bond angles between each, all lying in a plane perpendicular to the unhybridized p-orbital.
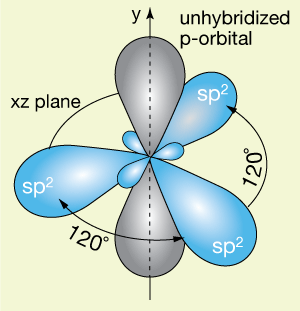
Let's consider the formation of a double bond (π-bond) between two sp2 hybridized carbon atoms. We begin with the two atoms separated like this.

Overlap of two of the sp2 orbitals, one from each atom, forms part of the bond. The other part comes from an interaction of the electrons in the unhybridized p-orbitals. As these are drawn nearer to each other, the molecular orbitals that form above and below the central sp2-sp2 bond create the rest of the π-bond.
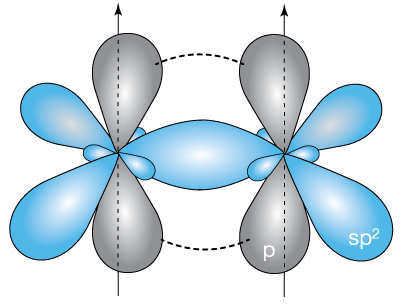
A more realistic impression of the orbitals of the full π-bond is shown below. This bonding arrangement, involving four total electrons, causes the molecule to be planar and rigid, with the C=C π-bond shorter than a C–C single (or sigma, σ) bond.
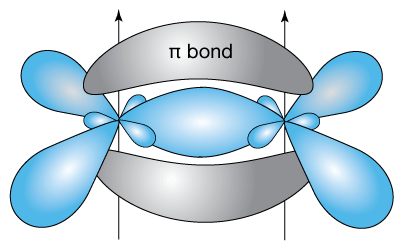
If we add four hydrogen atoms to each of the non-bonded sp2 orbitals still unoccupied in the drawing above, we get ethylene. Here is a simplified drawing of ethylene, C2H4, which has a C=C double bond with four hydrogen atoms bound to the other unpaired electrons (in sp2 hybrid orbitals) of the carbon atoms. Ethylene is a planar molecule.
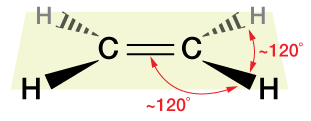
The C=C bond of ethylene, because of the way the molecular orbitals that form it are arranged, is stiff and does not rotate about the C=C axis. Thus ethylene is a fairly rigid planar molecule. Contrast that to ethane, C2H6, below, which has only sp3 hybridization and σ bonds.
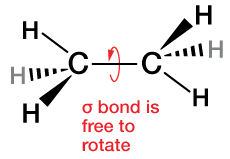
A typical C–C single bond is about 154 pm. The length of a double bond is about 20 pm (13%) shorter. Next we'll construct a triple bond, which will be even shorter, typically 120 pm for a C-C triple bond.
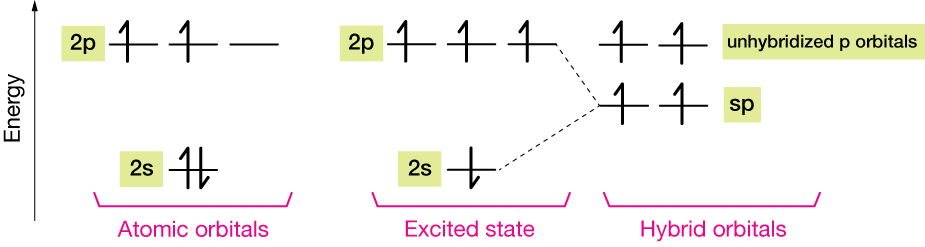
Triple bonding and sp hybridization
Well, we've seen hybrid orbitals of carbon formed from 1 part s and 3 parts p orbitals, and from 1 part s and two parts p orbitals. Why not 1:1 ?
This schematic diagram shows how sp (sp1, but we leave the 1 off) orbitals are formed.
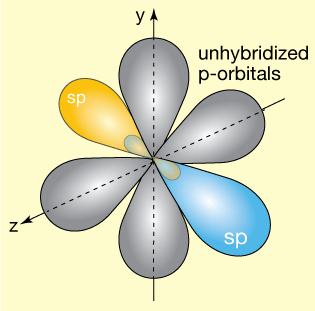
The resulting orbital arrangement looks like this. Two sp hybrid orbitals (yellow and blue in the figure) are orthogonal (perpendicular in both y & z directions, as shown) to two non-hybridized p-orbitals, each containing one electron.
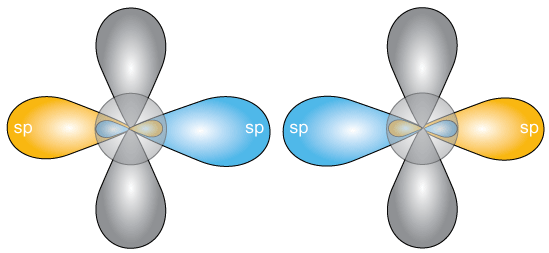
Now when two of these hybridized carbon atoms approach, two of the sp orbitals can form a molecular orbital and thus a bond,
Now in close proximity, the unhybridized p-orbitals of each atom can overlap and form their own molecular orbitals, a triple bond. Notice that in this drawing one unhybridized p-orbital of each carbon is seen end-on (gray circle).
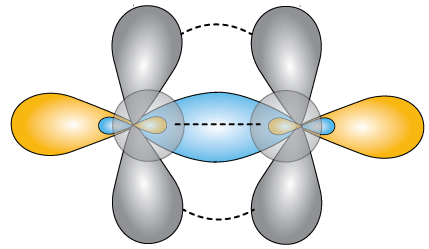
The complete picture of the bonding orbitals might look something like this. A central core of the merged sp orbitals is surrounded on two sides by pairs of molecular orbitals formed from the unhybridized p-orbitals. This is the nature of the triple bond. It occurs in carbon compounds and in the nitrogen molecule, N2, among others.
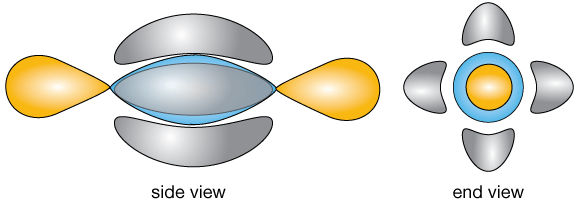
If we add two hydrogen atoms to bond with the remaining valence electron of each carbon atom, our completed molecular orbital diagram looks (very schematically) like this:
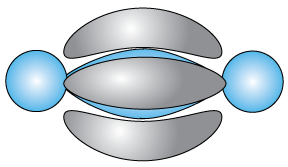
Finally, the ethyne molecule (or acetylene), C2H2, is a rigid, linear molecule,
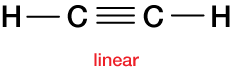
Examples of hybrid orbital systems
Nitrogen (N2)
The energy level diagram for the formation of the sp hybrid orbitals of the nitrogen atoms in N2 is shown below. There are five electrons in four orbitals before hybridization, and the same afterward. Notice that one of the sp hybrid orbitals is full; it contains two spin-paired electrons (up and down arrows).
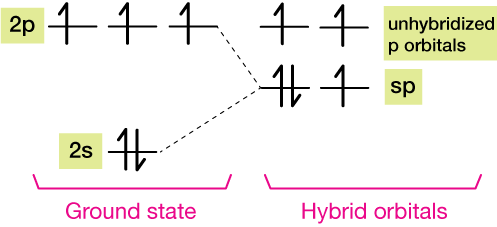
The hybridized and unhybridized orbitals look schematically like this. The yellow orbital is full, and won't form a bond. The other three orbitals are available for bonding, and we know from the Lewis structure that N2 forms a triple bond.
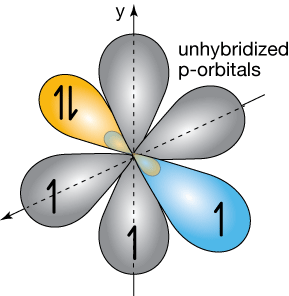
The figure below shows two hybridized nitrogen atoms lined up for bond formation. As in the last section, two of the unhybridized orbitals are meant to be poking into and out of the screen (gray circles in the middle).
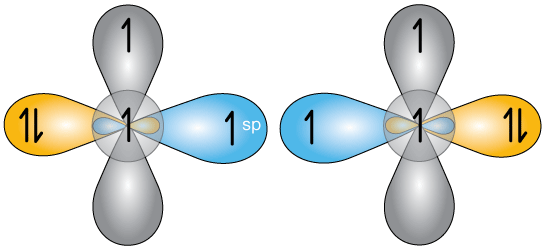
Finally, the complete bonding picture will look something like this.
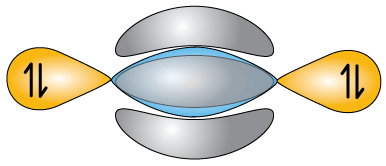
The stick drawing of N2 looks like this. I like to leave myself a reminder that there are lone pairs of electrons on either end, These will have an effect on the properties of the molecule.
![]()
The N≡N triple bond is one of the strongest bonds in nature, and leads to some interesting consequences in biology.
PCl5—sp3d hybridization
We can also have further levels of hybridization of orbitals, such as that found in the phosphorus atom of PCl5. In this molecule, the five ligands (Cl) all bond to equivalent sp3d orbitals, composed of one part s, three parts p and 1 part d orbital in the third (n = 3) shell.
For this to occur, we first consider an excited state in which one 3s orbital is excited into a 3d orbital:
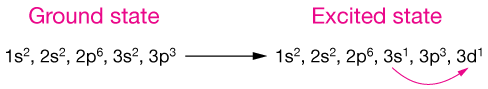
The hybridization of the five occupied orbitals looks like this. The energy levels aren't to scale; this is just a schematic picture.
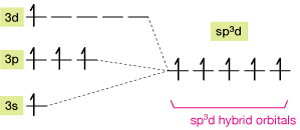
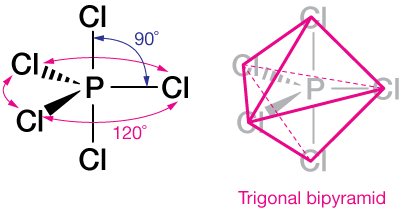
As predicted by VSEPR theory, those orbitals arrange themselves in a trigonal bipyramid, with three orbitals around the "equator" and two forming the "poles" of this nearly spherically-symmetric hybridized atom.

The molecule PCl5 looks like this. A trigonal bipyramid consists of two back-to-back three-sided pyramids.
Octahedral symmetry — sp3d2 hybridization.
A further orbital hybridization, sp3d2, consisting of one part s-orbital, three parts p-orbital and two parts d-orbital is possible, and leads to octahedral symmetry by forming six identical orbitals.
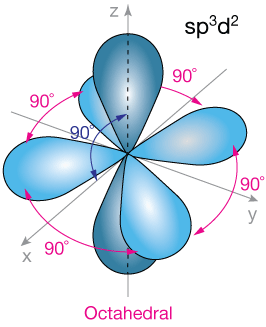
Examples of molecules with octahedral symmetry include SF6 and the ion [Co(NH3)6]3+

![]()
xaktly.com by Dr. Jeff Cruzan is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. © 2012, Jeff Cruzan. All text and images on this website not specifically attributed to another source were created by me and I reserve all rights as to their use. Any opinions expressed on this website are entirely mine, and do not necessarily reflect the views of any of my employers. Please feel free to send any questions or comments to jeff.cruzan@verizon.net.